Introduction
Cellular aging refers to the progressive decline in cellular function over time, driven by accumulated damage and changes in biological regulation.
This process affects all tissues and systems and underlies many of the functional changes associated with aging, including reduced regenerative capacity, increased inflammation, and impaired metabolic function.
Key Takeaways
- Cellular aging is driven by accumulated damage and regulatory changes
- Multiple biological processes contribute, including DNA damage and mitochondrial dysfunction
- Cellular senescence plays a key role in tissue aging
- Oxidative stress and inflammation accelerate cellular decline
- Aging is a systems-level process involving interconnected mechanisms
Why It Matters for Longevity
Longevity is not determined by a single organ or pathway, but by the cumulative function of cells across the body.
As cells lose efficiency, their ability to:
- Produce energy
- Repair damage
- Maintain structure
gradually declines.
This contributes to:
- Tissue dysfunction
- Increased disease risk
- Reduced physiological resilience
Understanding cellular aging provides a framework for addressing aging at its most fundamental level.
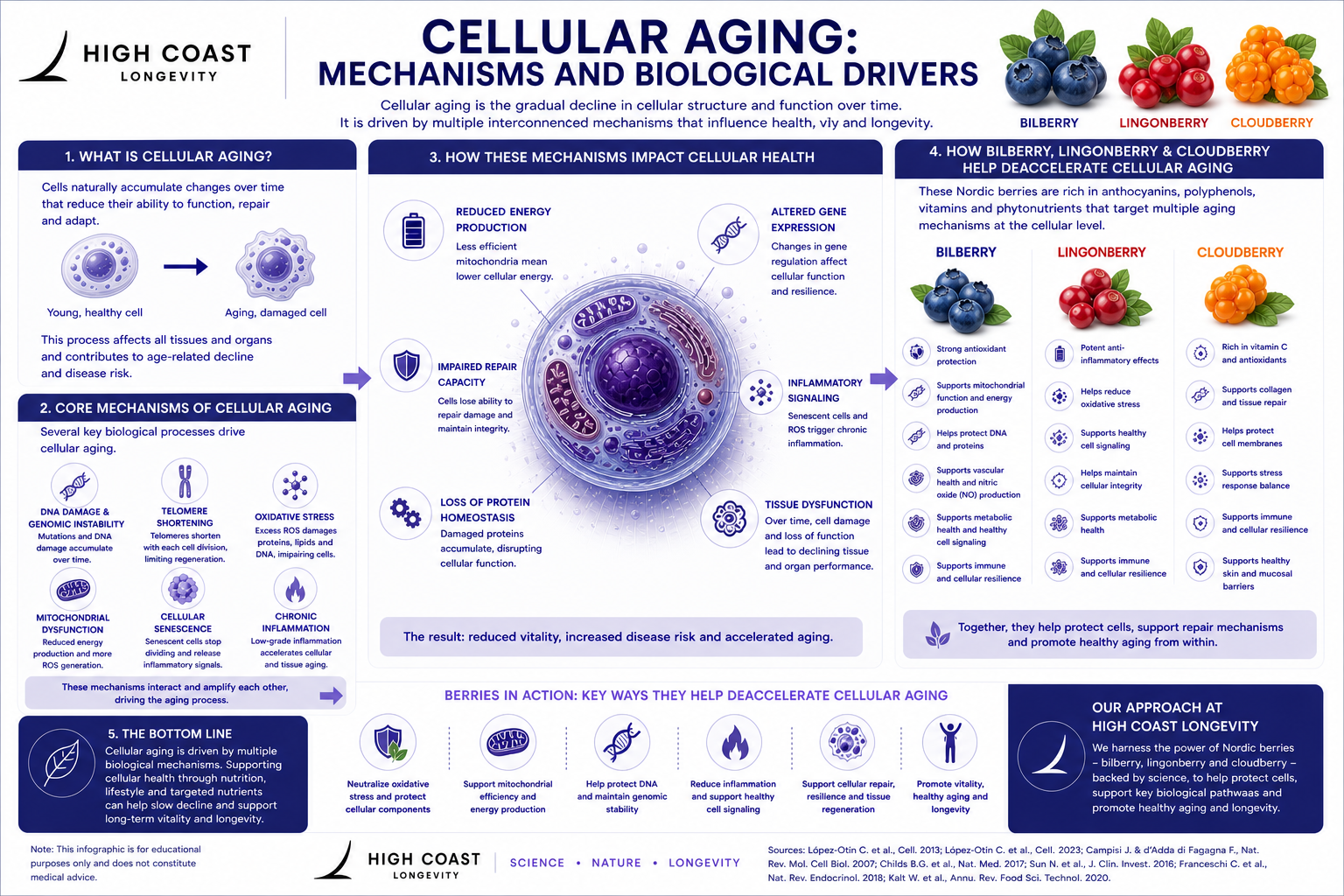
Biological Mechanisms
DNA Damage and Genomic Instability
Cells are continuously exposed to factors that can damage DNA, including:
- Reactive oxygen species
- Environmental stressors
- Replication errors
Over time, accumulated DNA damage can impair cellular function and increase the risk of malfunction or cell death.
Telomere Shortening
Telomeres are protective structures at the ends of chromosomes that shorten with each cell division.
When telomeres become critically short:
- Cells lose the ability to divide
- Cellular senescence is triggered
This limits tissue regeneration over time.
Cellular Senescence
Senescent cells are metabolically active but no longer divide.
They can:
- Accumulate in tissues
- Secrete inflammatory signals (SASP: senescence-associated secretory phenotype)
- Influence neighboring cells
This contributes to tissue dysfunction and chronic inflammation.
Mitochondrial Dysfunction
Mitochondria decline in efficiency with age, leading to:
- Reduced ATP production
- Increased oxidative stress
- Impaired metabolic regulation
This creates a feedback loop that accelerates cellular aging.
Oxidative Stress and Inflammation
Chronic oxidative stress and low-grade inflammation (“inflammaging”) are key drivers of cellular decline.
These processes:
- Damage cellular components
- Disrupt signaling pathways
- Promote senescence
What Accelerates Cellular Aging
- Chronic oxidative stress
- Inflammation
- Poor metabolic health
- Environmental stressors
- Sedentary lifestyle
- Nutritional deficiencies
What May Support Cellular Health
- Regular physical activity
- Balanced nutrition
- Adequate sleep and recovery
- Reduction of chronic inflammation
- Maintenance of metabolic health
These factors influence multiple aging pathways simultaneously.
Supporting Research
López-Otín et al. (2013)
Defined the “hallmarks of aging,” providing a framework for understanding the biological processes underlying cellular aging.
→ Cell
López-Otín et al. (2023)
Updated the hallmarks of aging, expanding the framework to include additional regulatory processes.
→ Cell
Campisi & d’Adda di Fagagna (2007)
Established the role of cellular senescence in aging and disease.
→ Nature Reviews Molecular Cell Biology
Childs et al. (2017)
Reviewed the impact of senescent cells on tissue function and aging.
→ Nature Medicine
Sun et al. (2016)
Explored mitochondrial dysfunction as a central component of aging.
→ Journal of Clinical Investigation
Franceschi et al. (2018)
Described “inflammaging” as a key driver of age-related decline.
→ Nature Reviews Endocrinology
Interpretation
Cellular aging is not driven by a single mechanism but by the interaction of multiple biological processes.
Damage accumulation, regulatory changes, and systemic factors combine to influence how cells function over time.
Interventions that support multiple pathways—rather than targeting a single mechanism—may be more effective in maintaining cellular function.
Connection to the High Coast Longevity Model
Within the High Coast Longevity framework, cellular aging represents the integrated outcome of multiple interacting systems.
It reflects:
- Mitochondrial efficiency
- Oxidative balance
- Nitric oxide signaling
- Cellular repair capacity
This positions cellular health as a central indicator of overall physiological resilience.
Part of the larger longevity framework
This article is one part of our broader review of aging biology, cellular resilience, vascular health, oxidative balance, and realistic nutritional strategies.
Read: Longevity Science Today
References
- López-Otín C et al. (2013). The hallmarks of aging. Cell.
- López-Otín C et al. (2023). Hallmarks of aging: An expanding universe. Cell.
- Campisi J, d’Adda di Fagagna F. (2007). Cellular senescence. Nat Rev Mol Cell Biol.
- Childs BG et al. (2017). Senescent cells in aging. Nature Medicine.
- Sun N et al. (2016). Mitochondrial dysfunction in aging. JCI.
- Franceschi C et al. (2018). Inflammaging. Nat Rev Endocrinol.