A deep dive into the 12 hallmarks of aging
Aging is not caused by one thing. It is not simply “wear and tear,” bad genes, slow metabolism, oxidative stress, inflammation, or declining hormones. Those are all pieces of the story, but none of them alone explains why a robust young organism gradually becomes vulnerable to frailty, cancer, cardiovascular disease, neurodegeneration, immune decline, sarcopenia, metabolic disease, and loss of regenerative capacity.
The best modern explanation is that aging is a systems-level failure of biological maintenance. Over time, damage accumulates in DNA, proteins, mitochondria, stem cells, immune signaling, the microbiome, and tissue architecture. The body does not merely collapse passively; it responds. But those responses, initially protective, can become maladaptive when they persist for decades.
This article is built around the modern hallmarks of aging framework. The original landmark review in Cell in 2013 proposed nine hallmarks of aging. The updated Cell review in 2023 expanded the framework to twelve hallmarks and emphasized that aging is not a linear chain of events, but an interconnected biological network.
The practical implication is profound: if aging is driven by identifiable biological mechanisms, then parts of aging may be measurable, modifiable, and eventually treatable.
Aging is a loss of biological resilience
A young body is not damage-free. DNA breaks occur, proteins misfold, mitochondria malfunction, inflammatory signals rise and fall, cells become stressed, and tissues suffer small injuries. The difference is that a young organism has extraordinary repair capacity. It detects damage, removes defective components, renews cells, restores tissue architecture, and re-establishes balance.
With aging, that capacity weakens. The body becomes less able to return to baseline after stress. A poor night of sleep, infection, surgery, psychological stress, metabolic overload, inactivity, or inflammation leaves a deeper biological footprint than it once did.
One useful way to understand aging is as a chronic, unresolved damage-response state. A 2025 Nature Communications perspective described aging as resembling a “wound that never starts healing,” arguing that aged organs show many features also seen after injury: inflammation, immune infiltration, cellular senescence, tissue remodeling, and altered repair.
This does not replace the hallmarks framework. It complements it. The hallmarks explain the molecular and cellular machinery. The unresolved-wound model explains what aging often looks like at the tissue level: the body is stuck between damage, defense, and incomplete repair.
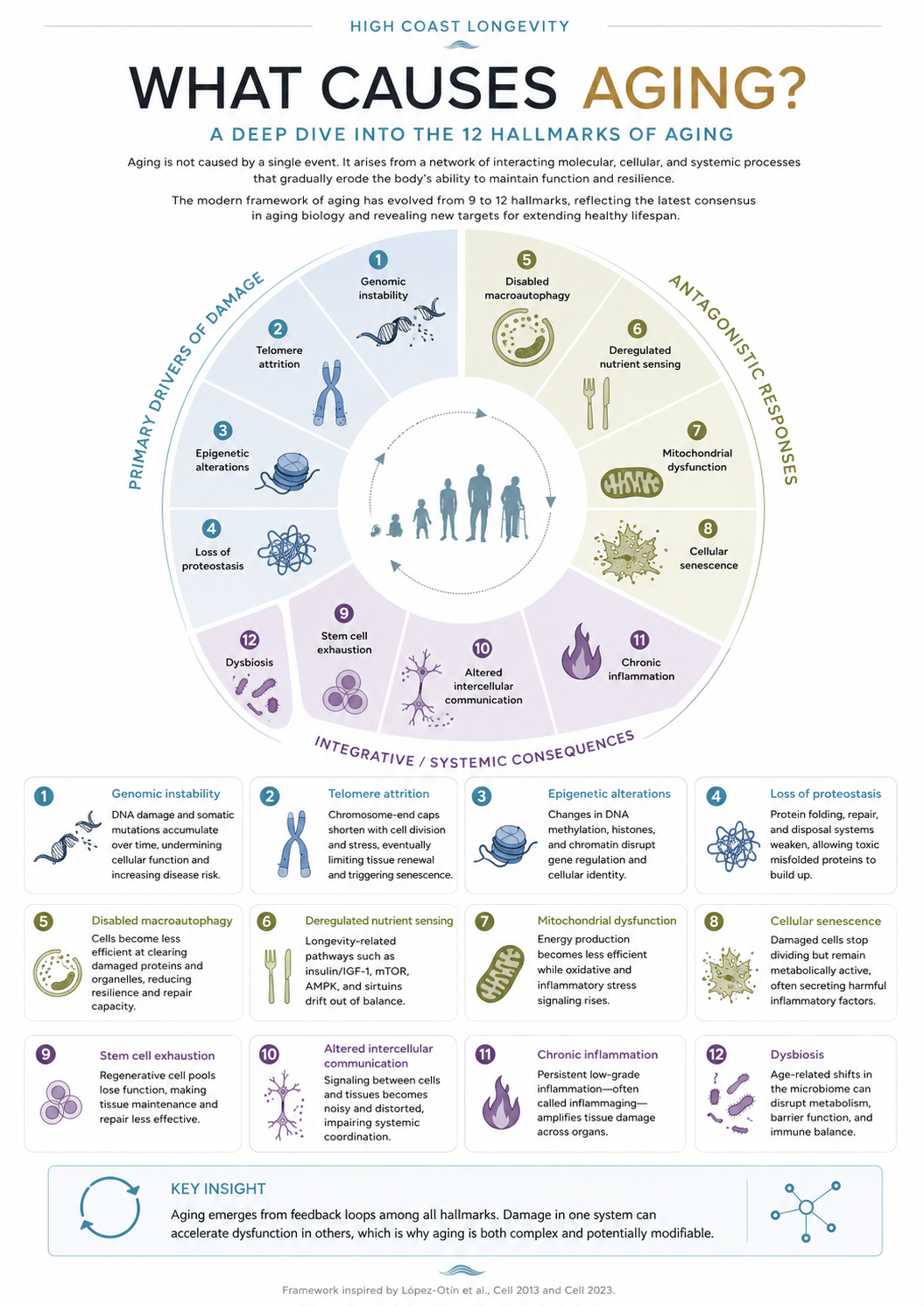
The 12 hallmarks of aging
The twelve hallmarks can be grouped into three broad categories:
Primary drivers of damage are the upstream sources of biological instability.
Antagonistic responses are protective mechanisms that become harmful when chronically activated or dysregulated.
Integrative and systemic consequences are the organism-wide failures that emerge when cellular and tissue systems lose coordination.
This grouping matters because aging is not simply “damage accumulation.” It is damage plus imperfect repair, plus maladaptive compensation, plus system-wide loss of communication.
1. Genomic instability: the slow corruption of the biological code
Every cell depends on DNA integrity. DNA contains the instructions for building proteins, maintaining cell identity, regulating metabolism, and coordinating repair. Yet DNA is under constant attack from replication errors, ultraviolet radiation, environmental toxins, reactive oxygen species, inflammation, and normal metabolic byproducts.
Cells have sophisticated DNA repair systems, but they are not perfect. Over time, mutations, chromosomal rearrangements, copy-number changes, mitochondrial DNA mutations, and other forms of genomic damage accumulate. This can impair cellular function, increase cancer risk, and trigger stress responses such as senescence or cell death.
Genomic instability is one of the primary hallmarks because it sits close to the origin of many downstream problems. When the genome becomes unstable, the cell must choose between risky survival, inflammatory senescence, programmed death, or malignant transformation.
One of the most important recent shifts in aging science is the recognition that the genome is not merely a static blueprint. It is constantly repaired, packaged, interpreted, and defended. Aging emerges not only when DNA is damaged, but when the systems that maintain genomic order gradually lose precision.
2. Telomere attrition: when chromosome ends become biological limits
Telomeres are protective caps at the ends of chromosomes. They help prevent chromosome ends from being mistaken for broken DNA. Each time many somatic cells divide, telomeres tend to shorten. Oxidative stress and inflammation can accelerate this process.
When telomeres become critically short, cells may stop dividing, enter senescence, or die. This limits tissue renewal, especially in organs that depend on ongoing cell replacement. Telomere attrition is therefore closely tied to stem cell exhaustion, immune aging, tissue frailty, and impaired healing.
However, telomeres are not simply “the cause of aging.” Some long-lived species do not fit a simplistic telomere-length model, and telomerase activation can carry cancer risk if not precisely controlled. The deeper point is that telomeres are part of a broader genome-protection system. They reveal how the body balances renewal against cancer suppression.
Too little renewal accelerates degeneration. Too much unchecked renewal increases cancer risk. Aging lives in that tension.
3. Epigenetic alterations: when cells forget who they are
If DNA is the hardware, the epigenome is part of the operating system. Epigenetic regulation determines which genes are turned on or off in a given cell. Liver cells, neurons, immune cells, muscle cells, and skin cells mostly contain the same DNA, but they behave differently because they use different gene-expression programs.
With age, DNA methylation patterns, histone modifications, chromatin structure, and higher-order genome organization change. These shifts can disturb cell identity and function. A cell may not become a different cell outright, but it can become less precise, less responsive, and less specialized.
This is why epigenetic clocks have become central to longevity research. They do not measure time directly; they measure molecular patterns that correlate with biological aging. A major 2023 Cell paper from the Biomarkers of Aging Consortium emphasized that validated biomarkers are essential for testing longevity interventions in humans, while also warning that the field still lacks full consensus on what makes a biomarker clinically reliable.
The promise is enormous. Epigenetic aging may be both a marker and a mechanism. But translating epigenetic reprogramming into safe human therapies remains one of the hardest challenges in medicine. Rejuvenating cells without triggering cancer, abnormal growth, or loss of identity will require extreme precision.
4. Loss of proteostasis: when cellular housekeeping breaks down
Proteins are the working machinery of life. They form enzymes, receptors, transporters, structural components, antibodies, signaling molecules, and molecular motors. But proteins must fold correctly, function correctly, and be removed when damaged.
Proteostasis means protein homeostasis: the ability of cells to maintain a healthy protein environment. This depends on chaperone proteins, the ubiquitin-proteasome system, autophagy, lysosomes, heat-shock responses, and other quality-control systems.
With age, proteostasis declines. Misfolded or damaged proteins accumulate. This is especially important in neurodegenerative disease, where protein aggregates are associated with Alzheimer’s disease, Parkinson’s disease, amyotrophic lateral sclerosis, and other disorders.
The problem is not merely the presence of damaged proteins. It is the declining ability to detect, refold, degrade, and recycle them. Aging cells increasingly lose the ability to keep their internal environment clean.
In practical longevity terms, proteostasis connects strongly to exercise, fasting biology, sleep, heat and cold stress, autophagy, mitochondrial function, and inflammation. Many interventions that appear “anti-aging” in model organisms may work partly because they improve cellular housekeeping.
5. Disabled macroautophagy: when the recycling system slows down
Autophagy is the cell’s recycling and renewal system. Macroautophagy, the best-known form, allows cells to engulf damaged proteins, dysfunctional organelles, and cellular debris, then deliver them to lysosomes for breakdown and reuse.
This process is essential for resilience. It helps cells survive nutrient stress, remove defective mitochondria, control inflammation, and maintain protein quality. When macroautophagy declines, cells become cluttered with damaged components. Mitochondria become more dysfunctional. Inflammatory signaling rises. Tissue repair becomes less efficient.
The 2023 hallmarks update recognized disabled macroautophagy as a distinct hallmark, reflecting how central cellular recycling is to aging biology.
This is one reason why interventions such as exercise, time-restricted feeding, caloric restriction, and certain pharmacological pathways attract so much attention in longevity science. They may influence autophagy-related biology. But it is important not to oversimplify. More autophagy is not always better. The goal is not maximum recycling; it is appropriate, context-sensitive recycling.
A young cell does not constantly destroy itself. It knows when to build and when to clean. Aging partly reflects the loss of that timing.
6. Deregulated nutrient sensing: when abundance signaling goes wrong
The body has ancient systems for sensing nutrients and energy availability. These include insulin and IGF-1 signaling, mTOR, AMPK, and sirtuins. Together, they help decide whether cells should grow, divide, repair, conserve energy, recycle components, or resist stress.
In youth, nutrient-sensing pathways are flexible. After feeding, the body builds and stores. During fasting, exertion, or scarcity, it shifts toward maintenance, repair, fat oxidation, and stress resistance.
With age and modern lifestyles, these pathways often become chronically distorted. Constant nutrient abundance, insulin resistance, visceral fat, inactivity, poor sleep, and inflammation can push the body into a state where growth signals remain high while repair signals become weaker.
This is one reason metabolic health is central to longevity. Glucose control, insulin sensitivity, muscle mass, mitochondrial fitness, and inflammatory tone all influence how nutrient-sensing pathways behave.
Deregulated nutrient sensing is also one of the most actionable hallmarks. Diet quality, protein distribution, resistance training, aerobic conditioning, sleep, circadian rhythm, and body composition all influence the metabolic environment in which aging unfolds.
But the key is balance. In a frail older person, aggressive fasting or protein restriction could worsen sarcopenia. In an insulin-resistant middle-aged person, improving metabolic flexibility may be powerful. Longevity medicine will increasingly need to personalize interventions rather than apply one universal rule.
7. Mitochondrial dysfunction: when power plants become stress signals
Mitochondria are often called the power plants of the cell, but that description is too narrow. They produce ATP, but they also regulate cell death, immune signaling, metabolic flexibility, redox balance, steroid synthesis, calcium handling, and inflammation.
With age, mitochondria often become less efficient. They may produce less usable energy, generate more reactive oxygen species, accumulate mutations in mitochondrial DNA, and send danger signals that activate inflammation.
Mitochondrial dysfunction is especially important because energy failure affects every tissue. The brain, heart, skeletal muscle, kidneys, immune system, and endocrine organs all depend heavily on mitochondrial performance.
But the old “free radical theory” of aging is too simplistic. Reactive oxygen species are not merely harmful exhaust. At controlled levels, they are signaling molecules that help cells adapt to exercise and stress. The problem is not oxidation itself; it is loss of redox control, impaired mitochondrial turnover, reduced mitophagy, and chronic stress signaling.
The most convincing human strategy for mitochondrial health remains physical activity, especially a combination of aerobic training and resistance training. Exercise stimulates mitochondrial biogenesis, improves insulin sensitivity, supports vascular function, and reduces inflammatory tone. In the future, mitochondrial-targeted drugs, NAD-related therapies, mitophagy activators, and cellular energetics interventions may become part of precision geromedicine, but the clinical evidence is still developing.
8. Cellular senescence: when damaged cells refuse to leave quietly
Cellular senescence is a state in which cells stop dividing but remain metabolically active. This can be protective. Senescence helps prevent damaged cells from becoming cancerous, supports wound healing, and participates in embryonic development.
The problem arises when senescent cells accumulate. Senescent cells can secrete inflammatory cytokines, chemokines, growth factors, and matrix-remodeling enzymes. This secretory profile, often called the SASP — senescence-associated secretory phenotype — can damage surrounding tissue, attract immune cells, disrupt stem cell niches, and amplify chronic inflammation.
In youth, senescent cells are usually cleared efficiently by the immune system. With age, production rises and clearance declines. The result is a growing burden of inflammatory, dysfunctional cells.
This hallmark has become one of the most exciting areas in longevity biotechnology. Senolytics are compounds designed to selectively eliminate senescent cells. Senomorphics aim to reduce harmful senescent signaling without killing the cells. Preclinical studies have shown striking effects in animal models, but human translation remains early and disease-specific.
The central lesson is that a process can be beneficial acutely and harmful chronically. Senescence is not “bad.” Persistent senescence is the problem.
9. Stem cell exhaustion: when regeneration loses depth
Stem cells maintain and repair tissues. They replenish blood, skin, intestinal lining, muscle, bone, and other organs. But stem cells do not operate in isolation. They depend on their niche: the local microenvironment of signals, immune cells, extracellular matrix, blood vessels, metabolism, and mechanical forces.
With age, stem cell pools can shrink, become less active, accumulate DNA damage, undergo epigenetic drift, lose mitochondrial fitness, or receive dysfunctional signals from aged tissue. The niche itself can become inflammatory, fibrotic, or metabolically hostile.
The result is slower repair and reduced tissue renewal. Skin becomes thinner. Muscle repair weakens. Blood production becomes less balanced. Immune renewal declines. The gut barrier may become more vulnerable. Bone remodeling changes. Reproductive tissues age.
A 2024 Nature Aging review on female reproductive aging illustrates how organ-specific aging involves both conserved hallmarks and tissue-specific mechanisms. The authors describe aging-related changes across the hypothalamic-pituitary-ovarian axis, ovarian stroma, egg quantity and quality, and uterine structure and function.
This is important because aging does not happen uniformly. Different tissues age at different rates, and the same hallmark can appear differently in the ovary, brain, immune system, muscle, skin, or cardiovascular system.
Stem cell rejuvenation is one of the great promises of longevity medicine, but also one of its greatest risks. Increasing regeneration without controlling cancer risk, fibrosis, immune signaling, and tissue architecture could be dangerous. The future will likely involve improving the stem cell environment as much as manipulating stem cells directly.
10. Altered intercellular communication: when the body loses coordination
Aging is not only a problem inside cells. It is also a problem between cells.
Cells constantly communicate through hormones, cytokines, neurotransmitters, extracellular vesicles, immune signals, metabolites, mechanical signals, and direct cell-cell contact. Organs also communicate with one another. Muscle talks to the brain. Fat talks to the immune system. The gut talks to the liver. The microbiome talks to the nervous system. Bone, pancreas, skin, blood vessels, and endocrine organs all participate in the network.
With age, this communication becomes noisier and less precise. Inflammatory signals may remain elevated. Hormonal rhythms flatten. Immune responses become both weaker and more inflammatory. Tissue repair signals become distorted. The nervous system becomes less adaptive. The extracellular matrix stiffens, changing mechanical signaling.
This hallmark explains why aging is systemic. A damaged tissue does not age alone. It sends signals that affect the whole organism. Visceral fat can promote inflammation. Dysbiotic gut microbes can alter immune tone. Senescent cells can disrupt neighboring cells. Muscle loss can worsen glucose control and reduce anti-inflammatory myokine signaling.
Aging is therefore not merely a collection of old cells. It is a communication failure across the biological network.
11. Chronic inflammation: inflammaging as an amplifier of decline
Inflammation is essential for survival. Without it, wounds would not heal and infections would become fatal. But inflammation is designed to be temporary. It should rise, solve a problem, and resolve.
With age, many people develop persistent low-grade inflammation, often called inflammaging. This can be driven by senescent cells, visceral adiposity, mitochondrial dysfunction, dysbiosis, chronic infections, damaged proteins, immune aging, leaky barriers, poor sleep, environmental exposures, and unresolved tissue stress.
Chronic inflammation is not just another hallmark. It is an amplifier. It worsens insulin resistance, damages blood vessels, disrupts stem cell niches, impairs brain function, accelerates muscle loss, promotes fibrosis, and increases risk for multiple age-related diseases.
This is why anti-inflammatory strategies are central to healthspan. But the goal is not to suppress the immune system broadly. That would increase infection and cancer risk. The goal is to restore immune resolution: strong defense when needed, rapid shutdown after the threat is gone, and low background inflammatory noise at baseline.
This distinction is crucial. Healthy aging does not mean a weak immune system. It means an immune system that is precise, responsive, and capable of returning to calm.
12. Dysbiosis: when the microbial ecosystem shifts against resilience
The gut microbiome is not separate from aging. It participates in metabolism, immune education, barrier function, inflammation, nutrient processing, bile acid metabolism, short-chain fatty acid production, and even communication with the nervous system.
With age, the microbiome often becomes less diverse and less resilient. Beneficial microbial functions may decline, while pro-inflammatory or opportunistic organisms may increase. Gut barrier integrity can weaken, allowing microbial products to stimulate systemic inflammation.
Dysbiosis connects directly to several other hallmarks. It can worsen chronic inflammation, alter nutrient sensing, affect mitochondrial function, influence immune aging, and disrupt intercellular communication. Conversely, diet, medications, infection, sleep, exercise, stress, and metabolic health all influence the microbiome.
This makes the microbiome both a cause and consequence of aging. It is not simply about “good bacteria” and “bad bacteria.” It is about ecosystem stability, metabolic output, immune balance, and host-microbe communication.
The most realistic near-term interventions are not exotic. They include dietary fiber diversity, fermented foods when tolerated, polyphenol-rich plants, metabolic health, reduced unnecessary antibiotic exposure, physical activity, and treatment of gastrointestinal pathology. More advanced microbiome therapeutics may eventually become important, but the field still needs stronger human evidence.
The feedback-loop model of aging
The twelve hallmarks are often presented as a list, but that can be misleading. In the body, they behave more like a network.
Genomic instability can trigger cellular senescence. Senescent cells can increase chronic inflammation. Chronic inflammation can damage mitochondria. Mitochondrial dysfunction can increase oxidative stress and inflammatory signaling. Dysbiosis can worsen inflammation and nutrient sensing. Impaired autophagy can worsen proteostasis and mitochondrial quality. Stem cell exhaustion reduces repair, which allows tissue damage to persist. Altered communication spreads local dysfunction throughout the organism.
This is why aging accelerates with time. It is not just that each hallmark slowly worsens. It is that the hallmarks begin to reinforce one another.
Aging is therefore a self-amplifying loss of resilience.
This also explains why single interventions rarely produce miraculous results. Targeting one hallmark may help, but if the broader network remains dysfunctional, the effect may be limited. The future of longevity medicine will likely involve combination strategies: metabolic optimization, exercise medicine, sleep and circadian repair, inflammation resolution, senescence targeting, microbiome support, mitochondrial health, regenerative medicine, and validated biomarkers.
Is aging a disease?
The phrase “aging is a disease” is controversial. Some argue that aging is universal, natural, and not itself a disease. Others argue that aging is the largest risk factor for nearly every chronic disease and has identifiable biological mechanisms, making it a legitimate target for medicine.
A more practical framing may be this: aging is not a single disease, but it is the biological substrate that makes many diseases possible.
Cardiovascular disease, Alzheimer’s disease, osteoarthritis, type 2 diabetes, cancer, frailty, osteoporosis, sarcopenia, macular degeneration, and immune decline do not appear randomly. Their risk rises dramatically with biological aging.
If medicine treats each age-related disease separately, it will always be late. Geroscience asks a more ambitious question: can we target the underlying biology of aging to delay or reduce multiple diseases at once?
A 2025 Cell review on geroscience and precision geromedicine describes how progress in aging biology is moving the field toward diagnostics and therapeutics designed to optimize health and prevent or treat age-related diseases across the lifespan.
This is the central promise of longevity medicine: not merely longer life, but longer function.
What actually accelerates aging?
The hallmarks describe biological mechanisms, but real life determines how strongly those mechanisms are activated. Several broad forces accelerate aging biology:
Metabolic dysfunction increases insulin resistance, inflammation, oxidative stress, vascular damage, and mitochondrial strain.
Physical inactivity reduces mitochondrial biogenesis, muscle mass, insulin sensitivity, vascular function, and anti-inflammatory signaling.
Poor sleep and circadian disruption impair metabolic regulation, immune function, hormone rhythms, brain clearance systems, and repair.
Chronic psychological stress can alter inflammatory tone, autonomic balance, sleep quality, glucose regulation, and health behavior.
Environmental exposures such as smoking, air pollution, ultraviolet radiation, toxins, alcohol excess, and poor diet increase DNA damage, inflammation, and tissue injury.
Visceral adiposity acts as an inflammatory and endocrine organ, worsening nutrient sensing and systemic communication.
Chronic infection or unresolved inflammatory disease keeps immune pathways activated.
Loss of social connection and purpose may indirectly influence aging through stress biology, behavior, sleep, inflammation, and neuroendocrine pathways.
These are not moral judgments. They are biological inputs. Longevity is not about perfection; it is about reducing chronic stress signals and increasing repair capacity.
What slows aging biology?
No intervention has yet been proven to comprehensively “reverse aging” in humans in the broad sense often claimed in marketing. But several strategies consistently support the biological systems that oppose accelerated aging:
Exercise remains the most evidence-aligned longevity intervention. It supports mitochondrial function, insulin sensitivity, vascular health, muscle mass, brain health, immune regulation, autophagy, and anti-inflammatory signaling.
Resistance training is essential because muscle is not only a movement organ; it is a metabolic and endocrine organ. Preserving muscle helps preserve glucose control, independence, injury resistance, and resilience.
Cardiorespiratory fitness is strongly associated with lower mortality risk and better functional aging. It reflects heart, lung, vascular, mitochondrial, and muscular integration.
Protein adequacy and nutrient density matter especially with age, when anabolic resistance makes muscle maintenance harder.
Metabolic health reduces stress on nutrient-sensing pathways and lowers inflammatory burden.
Sleep and circadian stability support hormonal rhythm, immune regulation, metabolic control, cognition, and repair.
Oral, gut, and skin barrier health may matter more than people think because barrier dysfunction can feed systemic inflammation.
Stress regulation and recovery help keep the nervous, endocrine, and immune systems from remaining in constant threat mode.
Precision prevention — measuring risk early and intervening before disease becomes advanced — is where longevity medicine may have its biggest near-term impact.
The point is not that lifestyle is the whole answer. It is not. But lifestyle controls many of the upstream inputs that shape the hallmarks.
The future: from anti-aging hype to precision geromedicine
The longevity field is moving through an important transition. The first phase was discovery: identifying the hallmarks, mechanisms, and model-organism interventions. The second phase was enthusiasm, sometimes mixed with hype. The next phase must be validation.
For the field to mature, it needs better answers to several questions:
Which biomarkers actually predict meaningful health outcomes?
Which interventions improve biological aging rather than merely changing a lab marker?
Which therapies are safe long-term?
Which interventions work for which people, at what age, and in what biological state?
How do we combine interventions without causing harm?
How do we extend healthspan without increasing cancer risk, frailty, immune suppression, or abnormal growth?
This is why biomarkers are crucial. The Biomarkers of Aging Consortium has emphasized that aging biomarkers are needed to evaluate longevity interventions over realistic timeframes, but also that standards and validation remain a major challenge.
The future is unlikely to be one magic pill. It will be a layered model: diagnostics, risk stratification, lifestyle medicine, preventive medicine, targeted pharmacology, regenerative approaches, and continuous monitoring.
That is the shift from “anti-aging” to precision longevity medicine.
Conclusion: aging is complex — but not mysterious
Aging is not caused by a single switch. It is caused by the gradual breakdown of biological maintenance across multiple levels: genome, epigenome, proteins, organelles, cells, tissues, immune signaling, metabolism, stem cells, and microbiome.
The twelve hallmarks give us a map:
- Genomic instability
- Telomere attrition
- Epigenetic alterations
- Loss of proteostasis
- Disabled macroautophagy
- Deregulated nutrient sensing
- Mitochondrial dysfunction
- Cellular senescence
- Stem cell exhaustion
- Altered intercellular communication
- Chronic inflammation
- Dysbiosis
But the deepest insight is not the list. It is the network.
Aging emerges when damage, stress responses, failed cleanup, impaired regeneration, inflammation, and systemic miscommunication begin to reinforce one another. That is why aging is so powerful. It is also why it may be modifiable.
The goal of longevity science is not immortality. It is to preserve the period of life when people can think clearly, move freely, resist disease, recover from stress, contribute, connect, and remain themselves.
In that sense, the question “What causes aging?” is more than a scientific question. It is the beginning of a new medical paradigm: one that treats healthspan as something measurable, protectable, and worthy of deliberate design.
Key scientific references with full links
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The Hallmarks of Aging. Cell. 2013;153(6):1194–1217.
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186(2):243–278.
- Biomarkers of Aging Consortium. Biomarkers of aging for the identification and evaluation of longevity interventions. Cell. 2023.
- Kroemer G, Maier AB, Cuervo AM, Gladyshev VN, Ferrucci L, Gorbunova V, Kennedy BK, Rando TA, Seluanov A, Sierra F, Verdin E, López-Otín C. From geroscience to precision geromedicine: Understanding and managing aging. Cell. 2025
- Ogrodnik M. Aging: the wound that never starts healing. Nature Communications. 2025;16:8732.
- Balough JL, Dipali SS, Velez K, Kumar TR, Duncan FE. Hallmarks of female reproductive aging in physiologic aging mice. Nature Aging. 2024;4:1711–1730.